HC Verma Class 11 Physics Solutions Chapter 14
HC Verma Solutions of Concept of Physics Part 1 Chapter -14 Some Mechanical Properties of Matter
HC Verma Solutions for Vol 1 & 2 – Free PDF Download
HC Verma Class 11 Physics Solutions Chapter 14: Some Mechanical Properties of Matter PDF
This page has detailed, step-by-step explanations of every question in HC Verma Class 11 Physics Solutions Chapter 14. For example, in the chapter “Some Mechanical Properties of Matter” which is the first chapter of Volume 1 of HC Verma’s concept of physics for jee, all of the questions are solved and the steps are explained to help you learn. Utopper is a smart way for students to go over the whole Physics Syllabus again and again. The questions and answers help them study in a way that will help them do well on their tests.
In the HC Verma Class 11 Physics Solutions Chapter 14 “Some Mechanical Properties of Matter” concept of physics, all of the questions are solved and the steps are explained to help you learn. On the Utopper website where students can get free Reference Book Solutions and other study materials like Revision notes, Sample papers, and Important Questions. Science will be easier to learn if you have access to HC Verma Solutions and solutions for other courses.
Click here to Buy Online – HC Verma Solutions Volume 1 Book
Here is a pdf of HC Verma Class 11 Solutions for Chapter 14: Some Mechanical Properties of Matter





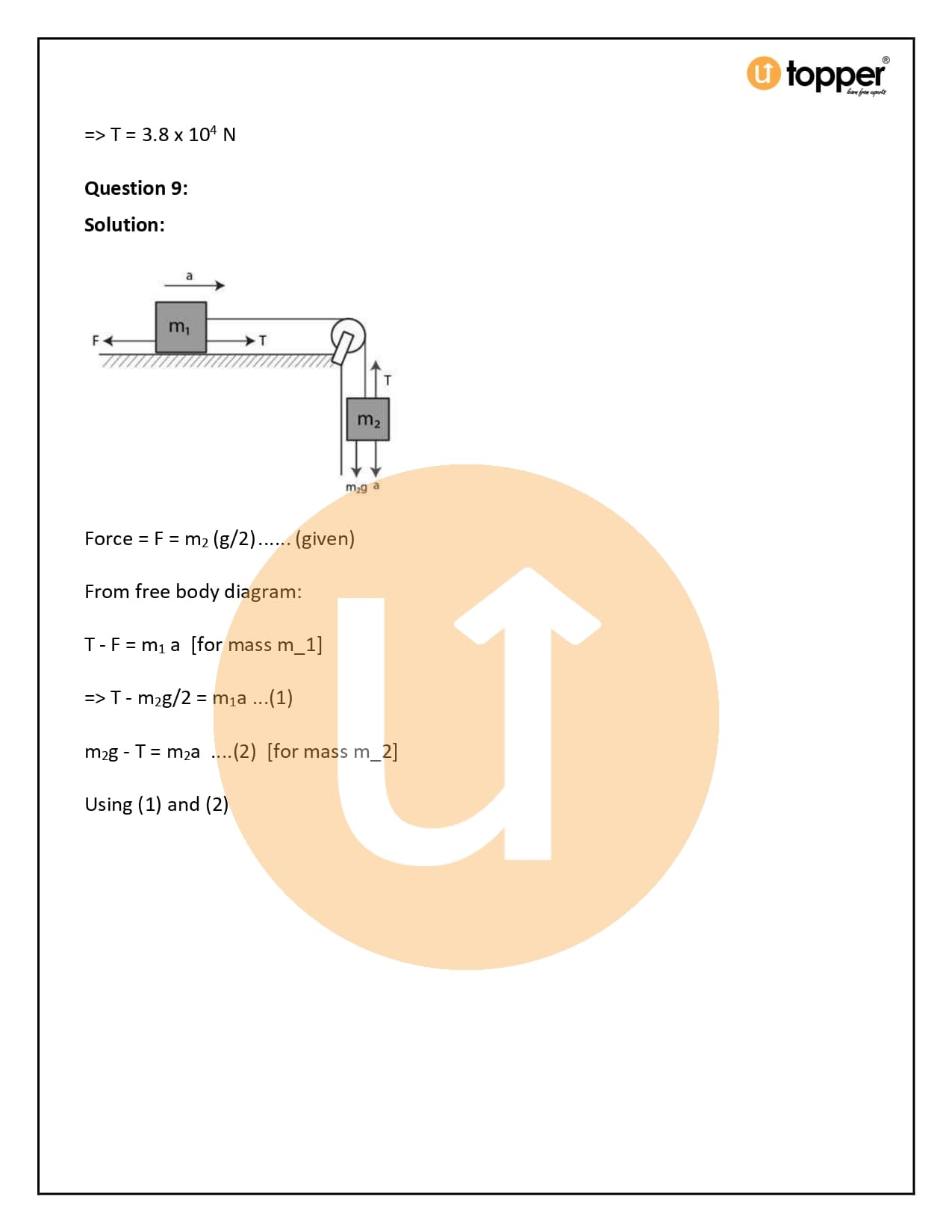

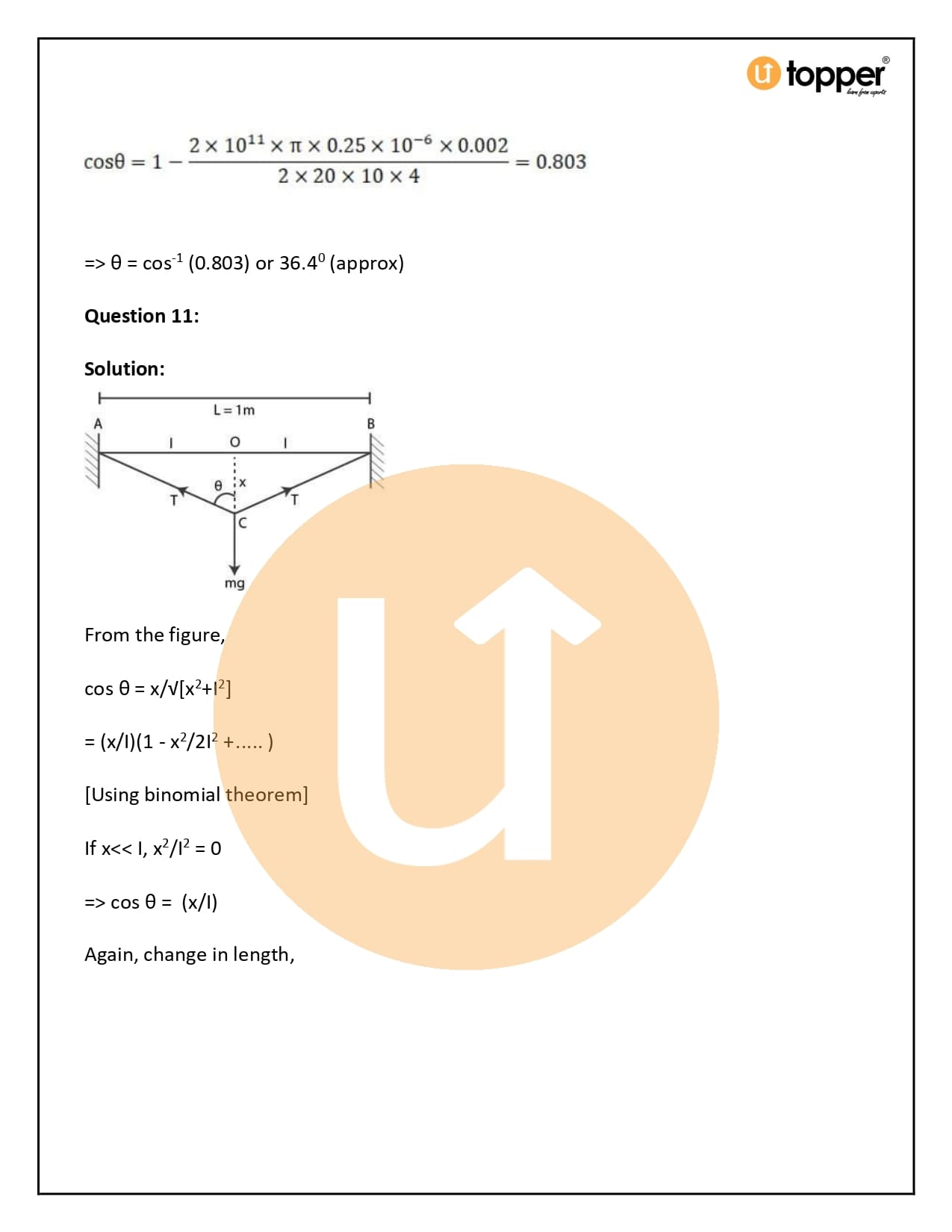














HC Verma Class 11 Physics Solutions
Chapter 1 – Introduction to Physics
Chapter 2 – Physics and Mathematics
Chapter 3 – Rest and Motion Kinematics
Chapter 4 – The Forces
Chapter 5 – Newton’s Laws of Motion
Chapter 6 – Friction
Chapter 7 – Circular Motion
Chapter 8 – Work and Energy
Chapter 9 – Centre of Mass, Linear Momentum, Collision
Chapter 10 – Rotational Mechanics
Chapter 11 – Gravitation
Chapter 12 – Simple Harmonics Motion
Chapter 13 – Fluid Mechanics
Chapter 14 – Some Mechanical Properties of Matter
Chapter 15 – Wave Motion and Waves on a String
Chapter 16 – Sound Waves
Chapter 17 – Light Waves
Chapter 18 – Geometrical Optics
Chapter 19 – Optical Instruments
Chapter 20 – Dispersion and Spectra
Chapter 21 – Speed of Light
Chapter 22 – Photometry

About the chapter: HC Verma Class 11 Solutions Chapter 14
Molecules and atoms are what make up matters. A nucleus and electrons make up an atom. Nucleons are the positive-charged protons and neutrons that make up the nucleus. The nucleus is made up of different nucleons that are held together by nuclear forces. Electromagnetic forces work between two electrons and between an electron and the nucleus. The way an atom is put together is because of these forces.
The structure of a molecule is determined by the forces between its atoms, and the structure of a material is determined by the forces between its molecules.
Some physical properties of matter
1. Intermolecular Force
- An Intermolecular Force is the force that acts between two molecules that are close to each other. This force can be felt by every molecule in a solid.
2. Elasticity
- The ability of the body to go back to its original shape and size is called “elasticity.” It’s usually because molecules and atoms have forces that pull them together. The Elastic Force is the name for this force.
3. Perfectly Elastic Body
- Perfectly elastic bodies go back to their original shape and size as soon as the forces that changed them are gone.
4. Plasticity
- It is the opposite of being flexible. The body can’t get back to its original size and shape.
5. Stress
- Stress is the force per unit area on a body’s surface when it changes shape. It is the force that works against the force from outside the body.
- Stress = F/ A
- F = Area, A = Force
- Stress is measured in Nm-2.
Stress Types :
- Normal Stress
- Tangential Stress
Normal Stress: Stress that is perpendicular to the surface of the body is normal stress. It also comes in two kinds:
- Tensile Stress
- Compressive Stress
Stress that acts parallel to the surface of the body is called tangential stress.
6. Young’s Modulus
- The ratio of longitudinal stress to longitudinal strain is called Young’s modulus. It stays the same for all materials.
7. Bulk Modulus
- The bulk modulus of an object is the ratio of its longitudinal stress to its volumetric strain.
8. Shear Modulus
- The ratio of the tangential stress to the shear strain is the shear modulus.
9. Poisson’s Ratio
Lateral strain is the difference between the change in diameter (D) and the diameter of the body at the start (D). Longitudinal strain is the difference between a person’s new length (l) and their original length (l). Here, Poisson’s ratio is the ratio of the sideways strain to the lengthwise strain.

Features of Utopper HC Verma Solutions for Class 11 Chapter 14
- Students can solve similar problems on their own with the help of HC Verma’s answers in the Utopper.
- Students are given answers that are correct and easy to understand.
- The solutions are given to match the level of understanding of a student in that class.
- The HC Verma solutions that Utopper gives the answer to and explains all of the questions in each chapter.
FAQ ( Frequently Asked Questions )
1. Where can I get HC Verma solutions?
Ans – Utopper offers HC Verma answers that are correct and have been worked out by experts. Our website, utopper.com, is where you can find HC Verma solutions. This is free, and if you study these answers, you can pass the JEE.
2. How Should I Study HC Verma For IIT JEE Preparation?
Ans – Students have used this book to study for JEE for the past twenty years, and they have all passed JEE. So, this book can’t be ignored. Once you’ve figured out how to solve the example problems, you can compare your answers to those in the book. If your answer is right, compare it to the solution to make sure you used the right steps to get there. If your answer is wrong, look at the other solutions to figure out “why” your answer is wrong. If you made a math or procedure mistake, you’ll need to find a way to fix it if you want to do well on an exam like the JEE.
3. Is HC Verma enough for the JEE Advanced?
Ans – Is the H.C. Verma book enough to prepare for the JEE Main and Advanced? H.C. Verma is a good book for getting ready for the JEE, and it goes into detail about each topic.
4. Who is better: SL Arora or HC Verma?
Ans – If you want to take the boards, SL Arora will do fine, and you won’t have to take HCV. But if you are studying for competitive exams like IIT/JEE mains or advanced, you should definitely go for HCV because it has more advanced problems and will help you solve more complicated problems.
