What is Acetaldehyde (CH3CHO)?
Acetaldehyde is a common, naturally occurring, odourless, combustible, and colourless liquid with a suffocating odour. Various plants, ripe fruits, vegetables, cigarette smoke, gasoline, and diesel exhaust contain acetaldehyde. This chemical is widely employed in the production of acetic acid, fragrances, dyes, and pharmaceuticals, as a flavouring agent, and as a metabolic intermediary in the metabolism of alcohol.
C2H4O is the chemical formula for the organic molecule acetaldehyde. Acute exposure to its fumes causes irritation of the eyes, skin, and lungs. It has an oppressive odour. It is non-corrosive to many metals, however, it has a narcotic effect and can irritate the mucous membranes. It is probable that acetaldehyde is a human carcinogen. It is also abbreviated by chemists as MeCHO where ‘Me’ means methyl.
Acetaldehyde Structure (C2H4O Structure)
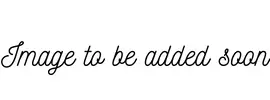
Acetaldehyde Molecular Formula
The formula for Acetaldehyde is CH3CHO or C2H4O.
Properties of Acetaldehyde – C2H4O
| C2H4O | Acetaldehyde |
| Molecular Weight/ Molar Mass | 44.05 g/mol |
| Density | 0.784 g/cm3 |
| Boiling Point | 20.2 °C |
| Melting Point | -123.5 °C |
| Solubility: | Soluble in water, ethanol, benzene, acetone, and slightly soluble in Chloroform |
Acetaldehyde IUPAC NAME
Ethanal is the IUPAC name for CH3CHO, also known as Acetaldehyde. It has two carbons, so “Eth” is used as a prefix, and it belongs to the aldehyde group, so “al” is used as a suffix.
Chemical Properties of Acetaldehyde – CH3CHO
1. Acetaldehyde reacts with Hydrazine
To make acetaldehyde phenylhydrazone, the nitrogen atom of phenylhydrazine serves as a nucleophile, attacking the electrophilic carbon of acetaldehyde and causing water to be removed.
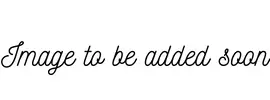
2. Acetaldehyde reacts with HCN
To create hydroxynitriles, hydrogen cyanide is injected over the carbon-oxygen double bond in aldehydes and ketones. For instance, with ethanal (an aldehyde), 2-hydroxypropanenitrile is produced.
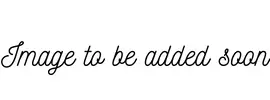
3. Acetaldehyde reacts with NaOH
When NaOH undergoes aldol condensation, acetaldehyde (A) reacts, generating beta-hydroxyacetaldehyde and, upon heating, alpha-beta-unsaturated aldehyde.
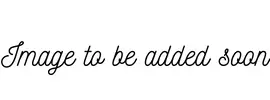
Acetaldehyde (C2H4O) Uses
- Used in the production of acetic acid.
- It is used in the production of resin.
- It is used to create disinfectants, medications, and fragrances.
- Used in the production of polyvinyl acetate.
- As a precursor, it is used to produce pyridine derivatives, crotonaldehyde, and pentaerythritol.
- In addition to being used as a preservative for fruit and fish in the production of vinegar, yeast, etc., acetaldehyde is also utilised as a preservative for meat and fish.
- It is employed as a solvent in a variety of industrial processes, including the production of paper, leather, and rubber.
- Additionally, it is utilised in the manufacture of n-butyraldehyde.
- It is used in leather tanning, as an alcohol denaturant, in fuel mixes, as a gelatin fibre hardener, in glue, and in casein products.
Acetaldehyde Toxicity
Ethanol’s first metabolite, acetaldehyde, is responsible for at least a portion of ethanol’s toxicity. Acetaldehyde is a substantially more potent toxin than ethanol. Some of the acetaldehyde enters the bloodstream, causing membrane damage and potentially scarring. In addition, it creates a hangover, a rapid heartbeat, a headache, and an upset stomach. Intoxication with acetaldehyde has the biggest effect on the brain. It inhibits memory and interferes with brain function.
Based on limited human cancer research and animal studies demonstrating nasal malignancies in rats and laryngeal tumours in hamsters, acetaldehyde is categorised as a possible human carcinogen. A substantial toxic metabolite, acetaldehyde, is one of the principal mediators of the fibrogenic and mutagenic effects of alcohol in the liver. The production of adducts is stimulated by acetaldehyde, which causes functional impairments in important proteins, such as enzymes, and DNA damage, which increases mutagenesis.
Frequently Asked Questions – FAQ
Q.1 What is acetaldehyde?
Various plants, mature fruits, vegetables, tobacco smoke, gasoline, and engine exhaust all contain acetaldehyde. This substance is extensively employed as a flavouring agent and a metabolic intermediary in the production of acetic acid, perfumes, dyes, and pharmaceuticals. CH3CHO is the chemical formula for acetaldehyde.
Q.2 What are the impacts of acetaldehyde on human health?
Acute (short-term) exposure to acetaldehyde causes irritation of the eyes, skin, and respiratory system. Chronic (long-term) acetaldehyde poisoning symptoms resemble those of alcoholism.
Q.3 What effect does acetaldehyde have on the liver?
Acetaldehyde, a significant toxic metabolite, is one of the principal mediators of the fibrogenic and mutagenic effects of alcohol in the liver. Acetaldehyde stimulates the development of adducts, which leads to functional impairments of essential proteins, such as enzymes, and DNA damage, which promotes mutagenesis.
Q.4 What is the difference between formaldehyde and acetaldehyde?
Iodoform will distinguish between formaldehyde and acetaldehyde. In response to a yellow precipitate, methyl ketones produce iodine and potassium hydroxide. With iodine and KOH, acetaldehyde bonds to carboxylic acid to form sodium salt. Iodoformity cannot be screened for with formaldehyde.
Q.5 Is acetaldehyde more toxic than alcohol?
Acetaldehyde is a significantly more potent toxin than ethanol, and at least a portion of ethanol toxicity can be attributed to acetaldehyde, the first metabolite of ethanol.
Q.6 Which foods include acetaldehyde?
Fruity-smelling acetaldehyde occurs naturally in foods such as fruits and fruit juice (0.2-230 ppm), vegetables (0.2-400 ppm), dairy products (0.001-76 ppm), and bread (4.2-9.9 ppm) 1), 2).
Q.7 What are the uses of acetaldehyde?
In other contexts, acetaldehyde functions as an addition, such as in fruit and fish preservatives, flavouring compounds, and gelatine hardening. Acetaldehyde is also employed in the production of vinegar, yeast, and as a preservative for fruit and seafood.
Q.8 Can acetaldehyde cause a positive iodoform test?
The iodoform test is a highly successful method for identifying whether an unidentified substance contains methyl ketone groups. CH3CHO is the chemical formula for acetaldehyde. Because it contains the COCH3 group (methyl ketone group), the iodoform test will be positive. Remember that it is the only aldehyde that produces a positive result on this test.

